Exeter Analytical - Model CE440 CHN Analyzer

PRINCIPLES OF OPERATION
 The samples to be analyzed are weighed into consumable tin capsules, which are then either crimped shut with tweezers or cold-welded shut if there are moisture or volatile sensitivities for the sample. Routine capsules (solids without sensitivities) are then placed inside an autosampler wheel where they are purged with helium, while non-routine capsules (goo, liquid, moisture/air-sensitive) are hand-dropped onto the sample ladle when prompted by the instrument.
The samples to be analyzed are weighed into consumable tin capsules, which are then either crimped shut with tweezers or cold-welded shut if there are moisture or volatile sensitivities for the sample. Routine capsules (solids without sensitivities) are then placed inside an autosampler wheel where they are purged with helium, while non-routine capsules (goo, liquid, moisture/air-sensitive) are hand-dropped onto the sample ladle when prompted by the instrument.
 The capsule is driven into a high temperature (1000°C) furnace and combusted in pure oxygen under static conditions. The tin capsules cause an initial exothermic reaction, raising the temperature of combustion to over 1800°C. At the end of the combustion period, a dynamic burst of oxygen is added to ensure total combustion of all inorganic and organic substances. The presence of silicon in a compound may necessitate the use of a combustion aid, to prevent the formation of silicon carbide.
The capsule is driven into a high temperature (1000°C) furnace and combusted in pure oxygen under static conditions. The tin capsules cause an initial exothermic reaction, raising the temperature of combustion to over 1800°C. At the end of the combustion period, a dynamic burst of oxygen is added to ensure total combustion of all inorganic and organic substances. The presence of silicon in a compound may necessitate the use of a combustion aid, to prevent the formation of silicon carbide.
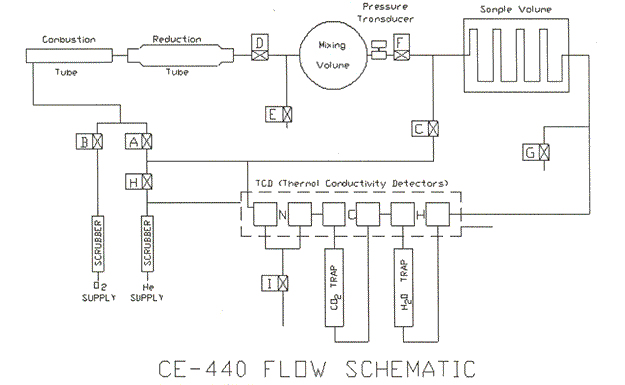
The resulting combustion products pass through specialized reagents to produce carbon dioxide (CO2), water (H2O) and Nitrogen (N2) and oxides of nitrogen. These reagents also remove other interferences including halogens, sulfur and phosphorus. The gases are then passed over copper to scrub excess oxygen and reduce oxides of nitrogen to elemental nitrogen. After scrubbing, the gases enter a mixing volume chamber to ensure a homogeneous mixture at constant temperature and pressure.
The mixture then passes through a series of high-precision thermal conductivity detectors, each containing a pair of thermal conductivity cells. Between the first two cells is a water trap. The differential signal between the cells is proportional to the water concentration, which is a function of the amount of hydrogen in the original sample. Between the next two cells is a carbon dioxide trap, with a similar process as above for the measuring of carbon. Finally, nitrogen is measured against a helium reference. The ashes from the combustion are scraped off of the ladle and the instrument is ready for the next run.
| ELEMENT | RANGE | DETECTION LIMIT | ERROR |
|---|---|---|---|
| C | 0-99% | 0.10% | 0.06% |
| H | 0-85% | 0.10% | 0.06% |
| N | 0-85% | 0.10% | 0.06% |
Source: Exeter Analytical, Inc